The loss of the protein pigment epithelium-derived factor, the study discovered, is a driver of aging-related changes in the retina.
Mice without a protective protein in their eyes have symptoms resembling age-related macular degeneration.
According to a recent National Eye Institute (NEI) study in mice, loss of the protein pigment epithelium-derived factor (PEDF), which protects retinal support cells, may promote age-related changes in the retina.
Age-related retinal diseases, such as age-related macular degeneration (AMD), can cause blindness since the retina is the light-sensitive tissue at the back of the eye. The new information could help develop medicines to stop AMD and other aging conditions of the retina. The research was published in the International Journal of Molecular Sciences. NEI is part of the National Institutes of Health.
“People have called PEDF the ‘youth’ protein because it is abundant in young retinas, but it declines during aging,” said Patricia Becerra, Ph.D., chief of NEI’s Section of Protein Structure and Function and senior author of the study. “This study showed for the first time that just removing PEDF leads to a host of gene changes that mimic aging in the retina.”
The retina is made up of layers of cells that work together to recognize and interpret light signals, which the brain utilizes to produce vision. The retina’s light-sensing photoreceptors are located atop a layer of support cells called the retinal pigment epithelium (RPE). When photoreceptors detect light, the RPE nourishes them and recycles “outer segments,” which get used up and their tips shed each time photoreceptors detect light.
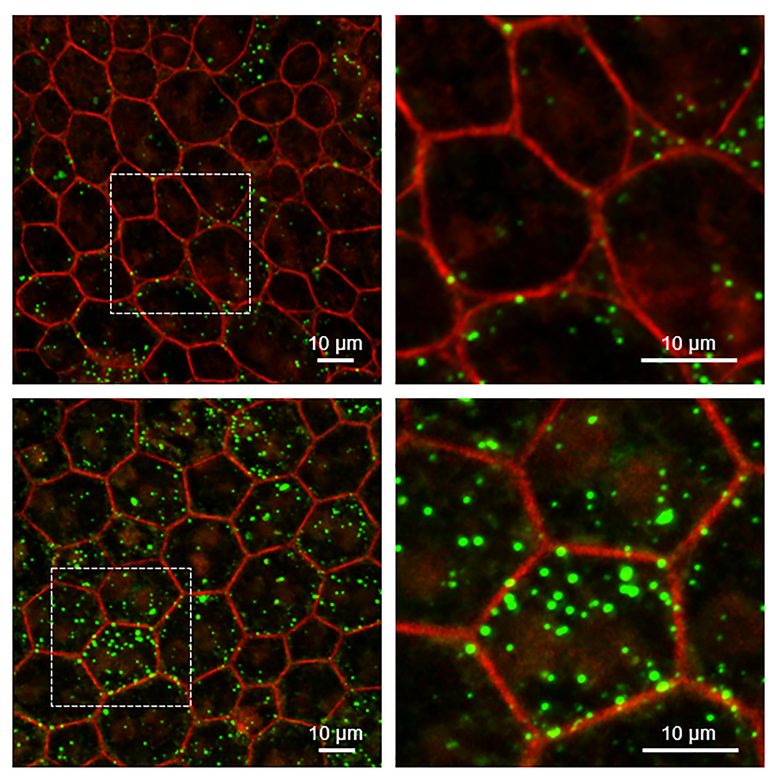
RPE from mice without Serpin1 accumulate more lipids than wild-type mice. Super-resolution confocal microscopy of RPE tissue from wild-type (upper) and Serpin1-null (lower) mice. Detailed images on the right are magnified regions of the RPE tissue imaged on the left (dotted square area). RPE cell boundaries are stained in red, and accumulated lipids are stained in green. Credit: Ivan Rebustini, NEI
Photoreceptor cells lose the capacity to create new segments and subsequently lose the ability to detect light if the RPE is unable to supply recycled components of older outer segment tips back to them. And without the nutrients that the RPE supplies, photoreceptors die. Senescence (aging) or death of RPE cells in the retina causes vision loss in individuals with AMD or certain types of retinal dystrophies.
Previous research from Becerra’s team and other groups have shown that PEDF shields retinal cells, protecting against both cellular damage and abnormal blood vessel growth in the retina. RPE cells produce and secrete the PEDF protein. The protein then binds to its receptor, PEDF-R, which is also expressed by RPE cells. Binding by PEDF stimulates PEDF-R to break down lipid molecules, key components of the cell membranes that enclose photoreceptor outer segments and other cellular compartments.
This breakdown step is a key part of the outer segment recycling process. And while researchers have known that PEDF levels drop in the retina during the aging process, it was not clear whether this loss of PEDF was causing, or merely correlated with, age-related changes in the retina.
To examine the retinal role of PEDF, Becerra and colleagues studied a mouse model that lacks the PEDF gene (Serpin1). The researchers examined the cellular structure of the retina in the mouse model, finding that the RPE cell nuclei were enlarged, which may indicate changes in how the cells’ DNA is packed.
The RPE cells also had turned on four genes associated with aging and cellular senescence, and levels of the PEDF receptor were significantly below normal. Finally, unprocessed lipids and other photoreceptor outer segment components had accumulated in the RPE layer of the retina. Similar changes in gene expression and defects in RPE metabolism are found in the aging retina.
“One of the most striking things was this reduction in the PEDF receptor on the surface of the RPE cells in the mouse lacking the PEDF protein,” said the study’s lead author, Ivan Rebustini, Ph.D., a staff scientist in Becerra’s lab. “It seems there’s some sort of feedback-loop involving PEDF that maintains the levels of PEDF-R and lipid metabolism in the RPE.”
While at first glance, the retinas of these PEDF-negative mice appear normal, these new findings suggest that PEDF is playing a protective role that helps the retina weather trauma and aging-related wear and tear.
“We always wondered if loss of PEDF was driven by aging, or was driving aging,” said Becerra. “This study, especially with the clear link to altered lipid metabolism and gene expression, indicates the loss of PEDF is a driver of aging-related changes in the retina.”
Reference: “PEDF Deletion Induces Senescence and Defects in Phagocytosis in the RPE” by Ivan T. Rebustini, Susan E. Crawford and S. Patricia Becerra, 13 July 2022, International Journal of Molecular Sciences.
DOI: 10.3390/ijms23147745
The study was funded by the National Eye Institute.
scitechdaily.com